updated September 20. 2025
This information is current as of the date of original publication or update but may have changed by the time you read this. Do not use this information for diagnosis or treatment purposes. Before making health decisions, discuss with a qualified healthcare professional.
Kennedy visits the Senate
On September 4, 2025, HHS Secretary Robert Kennedy Jr. appeared before the Senate Finance Committee and was heatedly questioned about several issues. Most of his testimony centered around his firing of the newly confirmed CDC Director Dr. Susan Monarez.
He was also grilled on his seeming lack of support for vaccination as an important public health measure. They questioned why he appointed several antivaccine scientists to the Advisory Committee on Vaccines. Even the Republican Senators who had voted for his appointment expressed frustration with his leadership.
In this post I explain some of what lead to this confrontation.
Trump appoints a CDC Director
After being without a CDC Director since he took office in January 2025, President Trump and the Senate confirmed Dr. Susan Monarez. This is the first time in history that a CDC Director needed Senate approval.
President Trump posted this on social media about his choice for CDC director.
“Dr. Monarez brings decades of experience championing Innovation, Transparency, and strong Public Health Systems. She has a Ph.D. from the University of Wisconsin, and PostDoctoral training in Microbiology and Immunology at Stanford University School of Medicine.
“As an incredible mother and dedicated public servant, Dr. Monarez understands the importance of protecting our children, our communities, and our future. Americans have lost confidence in the CDC due to political bias and disastrous mismanagement.
Dr. Monarez will work closely with our GREAT Secretary of Health and Human Services, Robert Kennedy Jr. Together, they will prioritize Accountability, High Standards, and Disease Prevention to finally address the Chronic Disease Epidemic and, MAKE AMERICA HEALTHY AGAIN!”

That was in July 2025. Something changed by late August.
I share this article published by KFF Health News. It was written by Dr. Celine Gounder, a physician and medical journalist who specializes in infectious diseases and global health. She was a member of the COVID-19 Advisory Board of President Joe Biden.
(I have edited the article slightly for length.)
Senior CDC Officials Resign After Dr. Monarez’s Ouster, Citing Concerns Over Scientific Independence
Written by Dr. Céline Gounder August 29, 2025
Four senior officials with the CDC, Centers for Disease Control and Prevention, announced their resignations in recent days, citing what they described as growing political interference in the agency’s scientific work, particularly regarding vaccines.
Two of them — Dr. Debra Houry, the CDC’s chief science and medical officer, and Dr. Demetre Daskalakis, who led the National Center for Immunization and Respiratory Diseases — stepped down on Aug. 27, hours after the White House announced the firing of CDC Director Susan Monarez.
Dr. Monarez, confirmed by the Senate in late July, was removed less than a month into her tenure.
White House spokesperson Kush Desai said Monarez “was not aligned with the President’s agenda of Making America Healthy Again.” Monarez’s attorneys argue that the dismissal is unlawful, as only the apresident can remove a Senate-confirmed director.
On Aug. 28, Jim O’Neill, the deputy secretary of the Department of Health and Human Services, was chosen to serve as acting CDC director. In an internal email sent to CDC staffers Secretary Robert F. Kennedy Jr. confirmed O’Neill as the acting CDC director without addressing Monarez’s departure.
“I am committed to working with you to restore trust, transparency, and credibility to the CDC,” Kennedy told CDC employees, later writing that
“President Trump and I are aligned on the commonsense vision for the CDC: Strengthen the public health infrastructure by returning to its core mission of protecting Americans from communicable diseases by investing in innovation to prevent, detect, and respond to future threats.”
HHS Secretary Robert Kennedy, Jr.
Concern for Vaccine Policy
Houry and Daskalakis said they had become increasingly uneasy about how vaccine policy was being handled. Both pointed to preparations for the Advisory Committee on Immunization Practices, ACIP, meeting, which recommends vaccine schedules.
Houry said they feared “some decisions had been made before there was even the data or the science to support those. We are scientists, and that was concerning to us.”
Daskalakis added that he was “very concerned that there’s going to be an attempt to relitigate vaccines that have already had clear recommendations with science that has been vetted,” which he warned could undermine public trust. “If you can’t attack access, then why not attack trust? And that’s what I think the playbook is,” he said.
“there’s going to be an attempt to relitigate vaccines that have already had clear recommendations with science that has been vetted,
If you can’t attack access, then why not attack trust? And that’s what I think the playbook is,”
Dr. Demetre Daskalakis
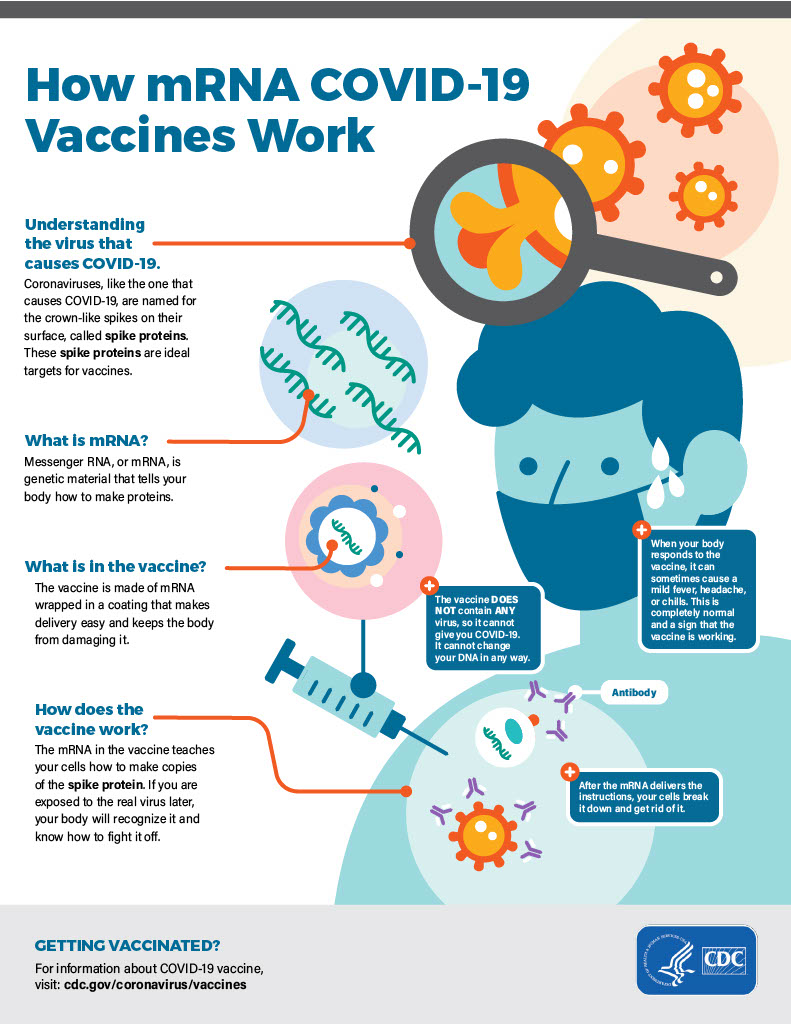
Lack of communication and transparency
Both officials cited instances in which evidence reviews were altered or withdrawn. A CDC analysis of thimerosal, a vaccine preservative, was briefly posted before being taken down at the HHS’ direction.
“If there’s something that doesn’t line up with the recommendations, then that information will be taken down, and it’s not there for the public to see for openness and transparency,” Houry said.
The two also criticized the lack of direct communication between CDC scientists and HHS leadership. Daskalakis said his team was never invited to brief Kennedy on measles and COVID-19.
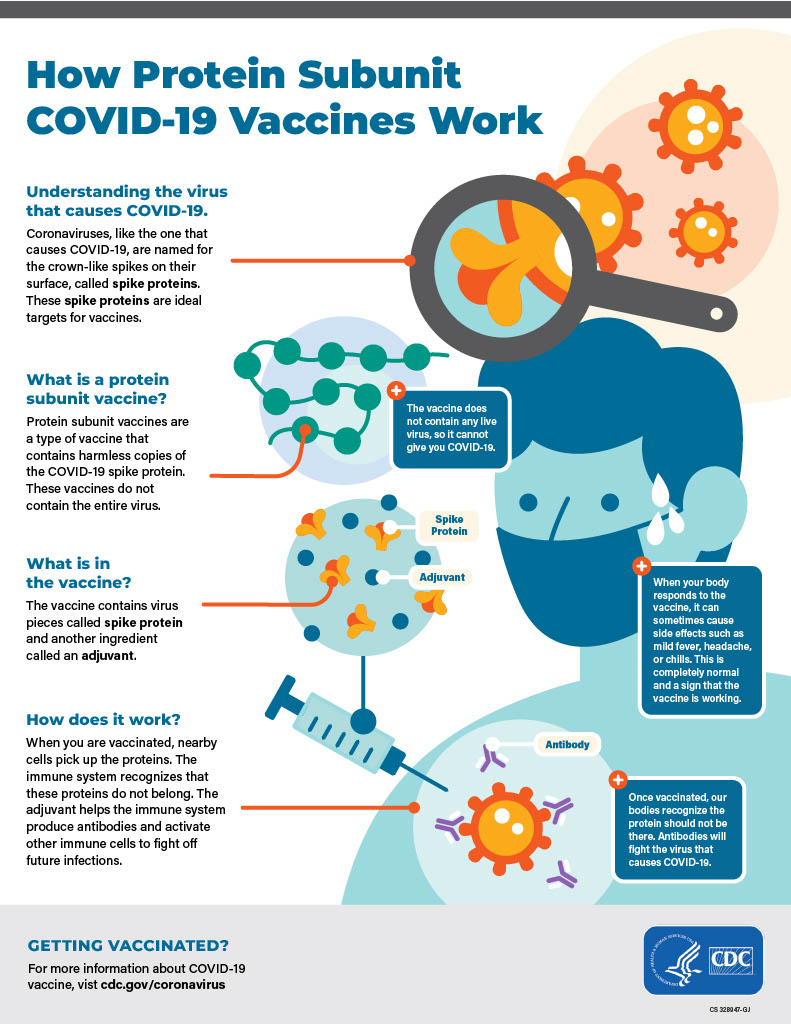
When asked about Kennedy’s calls for “radical transparency,” Houry and Daskalakis learned about changes to the COVID-19 vaccine schedule for children through social media, not through internal channels.
“The radical transparency manifested itself by a Twitter post, which is how Dr. Houry and I learned that the secretary had mandated the change in the children’s vaccine schedule for COVID,” Daskalakis recalled.
“What is the background that led to that decision? And we were denied access to that information. So, I don’t think that that’s radically transparent,” Daskalakis said.
CBS News and KFF Health News reached out to HHS for comment on some of the allegations made by Houry and Daskalakis but did not immediately hear back.
Scientists Raise An Alarm for Public Health
Both officials said they had no jobs lined up when they resigned. Houry described the decision as an effort to raise the alarm about the direction of the agency.
“For us, this was really sending out a bat signal,” Houry said. “We were the very senior scientists and career leaders at CDC. We thought this was the time to stand together and try to do what we could to raise the alarm around public health in our country.”
Daskalakis said remaining at the CDC under current conditions would have made them complicit in what he called the “weaponization” of public health.
“The safety has already been compromised. … We are flying blind in the U.S. already. If we continued … we would be complicit and would be facilitating the ability to go from flying blind to actively harming people,” he said.
Houry emphasized the severity of the moment by noting that she left without a backup plan.
“My leaving without a job was really just showing how dire the circumstances had become,” Houry said.
Daskalakis said his decision was also shaped by his medical oath.
“As a physician, I take the Hippocratic oath: First, do no harm. I am seeing ideology permeating science in a way that is going to harm children and adults. … I think we are seeing things that are happening that are making our country less prepared to be able to respond to the everyday pathogens … but also … to the next big thing.”
Both also expressed concerns about their personal safety in the current climate.
“The environment we live in … stoked by misinformation, especially from people considered by some to be health authorities, makes me worried for all of us in public health,” Daskalakis said. “I am concerned, but that’s part of our job … to be brave and continue to speak the truth even when we are outside of the CDC.”

Gun Violence-Another Public Health Issue
The resignations came weeks after a shooting outside the CDC’s Atlanta headquarters, which law enforcement linked to COVID misinformation.
Houry said the White House response to the shooting was muted. Kennedy toured the site but later gave an interview expressing distrust of experts.
“That was after the attack. It was based on COVID misinformation. So this is when we were trying to build trust,” she said.
Daskalakis added that while Kennedy later described mass shootings as a public health crisis, he believed the secretary should address misinformation as a root cause.
“The misinformation about the COVID-19 vaccine — that has been documented by the Georgia Bureau of Investigation” as the reason for the CDC shooting. “I would really recommend that the secretary actually do take his own advice and actually address the core problem that led to that shooting as well,” he said.
He also noted that the CDC’s gun violence prevention programs had been sharply reduced. “We talk about violence as a public health problem.
It is, and there’s things we can do to prevent it. Unfortunately, the majority of that program, the staff are terminated,” he said.
“We talk about violence as a public health problem.
Dr. Daskalakis
It is, and there’s things we can do to prevent it. Unfortunately, the majority of that program, the staff are terminated,”
The firings and resignations have sparked calls for oversight. Independent Sen. Bernie Sanders of Vermont called for a bipartisan investigation, Democratic Sen. Patty Murray of Washington urged Kennedy’s removal, and Republican Sen. Bill Cassidy of Louisiana — who voted to confirm Kennedy’s appointment as HHS secretary — said the developments would “require oversight.”
The events come as the FDA narrowed eligibility for updated COVID-19 vaccines to older adults and people with risk factors for severe COVID.
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF—an independent source of health policy research, polling, and journalism. Learn more about KFF.
Subscribe to KFF Health News’ free Morning Briefing.
This article first appeared on KFF Health News and is republished here under a Creative Commons Attribution-NoDerivatives 4.0 International License.![]()
Dr. Monarez Speaks to the Senate
On September 17, 2025, “She testified that she was ousted last month because she refused to cede to Health Secretary Robert F. Kennedy Jr.’s demands to pre-approve vaccine recommendations for the public and fire career scientists.
“He just wanted blanket approval,” Monarez told members of the Senate Committee on Health, Education, Labor and Pensions on Wednesday. “Even under pressure, I could not replace evidence with ideology.”
Monarez described a string of events that she said eventually led to her dismissal, culminating with a “tense” meeting in late August when she clashed with Kennedy over his plans for an influential CDC committee that issues recommendations on vaccines.”
Read the full report from NPR at this link
Ousted CDC director testifies she was fired for resisting pressure from RFK Jr.
Learn about the CDC
I hope you will take the time to learn more about the Centers for Disease Control and Prevention. According to the website, the
- CDC is the nation’s leading science-based, data-driven, service organization that protects the public’s health.
- CDC puts science into action to help children stay healthy so they can grow and learn; to help families, businesses, and communities fight disease and stay strong; and to protect the public’s health.
The professionals at the CDC cannot do their job alone. They need the support of elected and appointed government officials and the people they serve.
Health and safety threats are often new and unpredictable, like a viral pandemic or natural disaster, and multiple agencies need to work together to successfully manage threats. Cooperation and support are needed. Blaming and accusations are unproductive and unprofessional and contribute to lack of trust by the people they serve.
Post Graphics
The two graphics about vaccines are currently on the CDC website. The cover photo of CDC headquarters is also on the site.
Exploring the HEART of Health
I’d love for you to follow this blog and follow me on social media.
I share information and inspiration to help you transform challenges into opportunities for learning and growth.
Add your name to the subscribe box to be notified of new posts by email. Click the link to read the post and browse other content. It’s that simple. No spam.
I enjoy seeing who is new to Watercress Words. When you subscribe, I will visit your blog or website. Thanks and see you next time.

Dr Aletha